Medical
We help medical device companies along their entire supply chain, from new product design & engineering to molding, assembly, and packaging.
Injection Molding
13485 Certified. 9001 Certified. FDA Registered. Clean Room Molding & Assembly. Medical Grade Resins.
Dip Molding
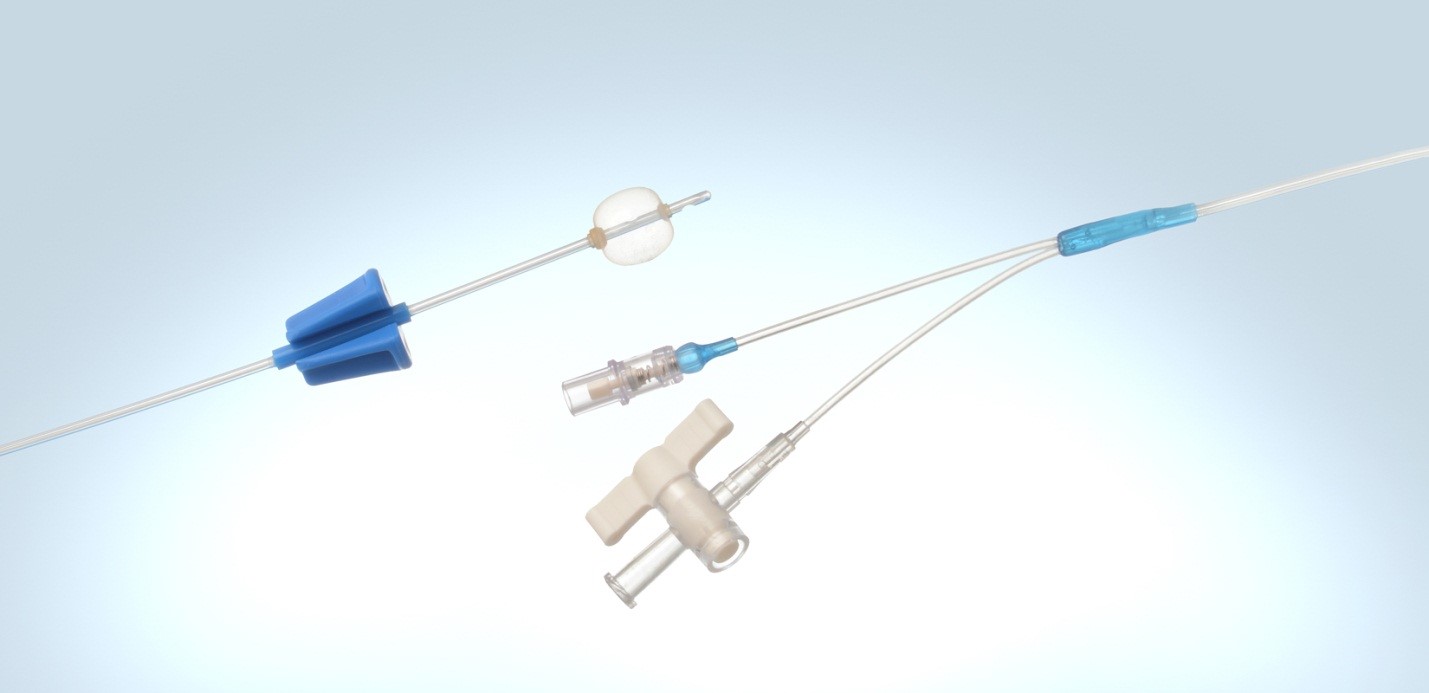
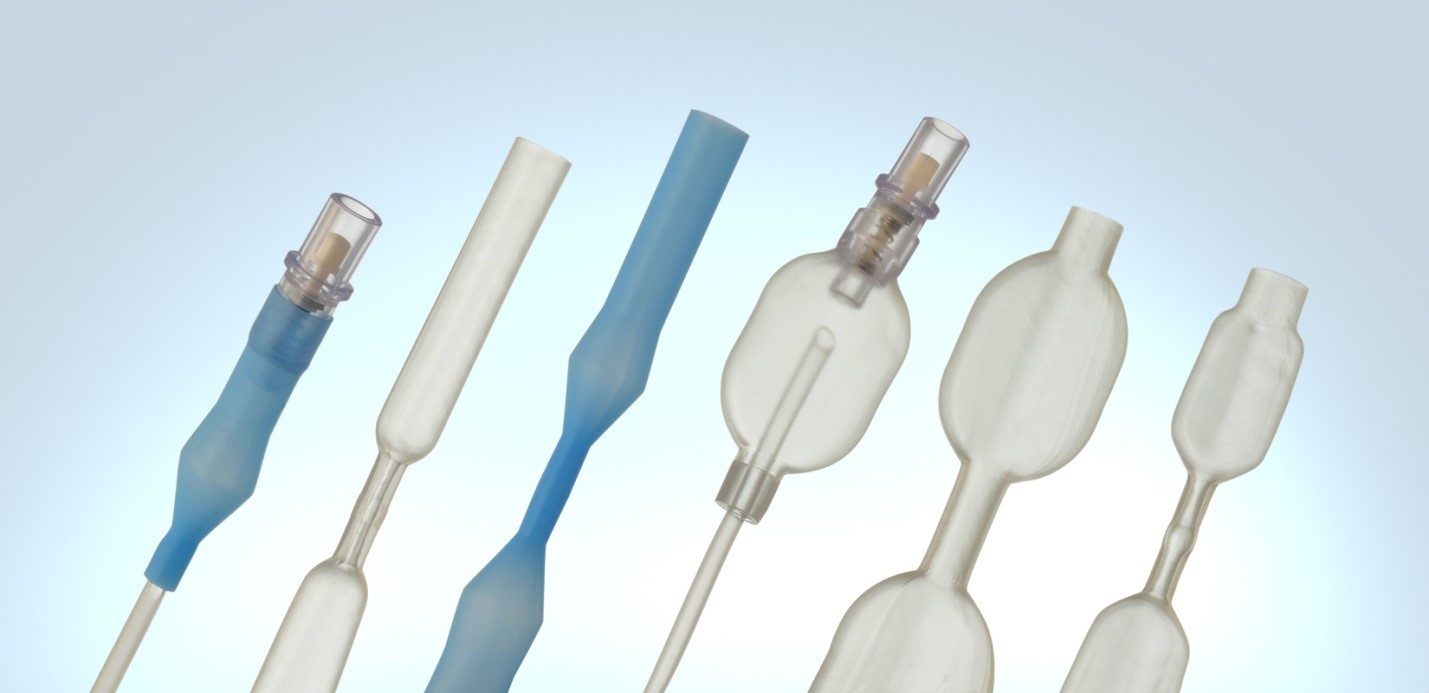
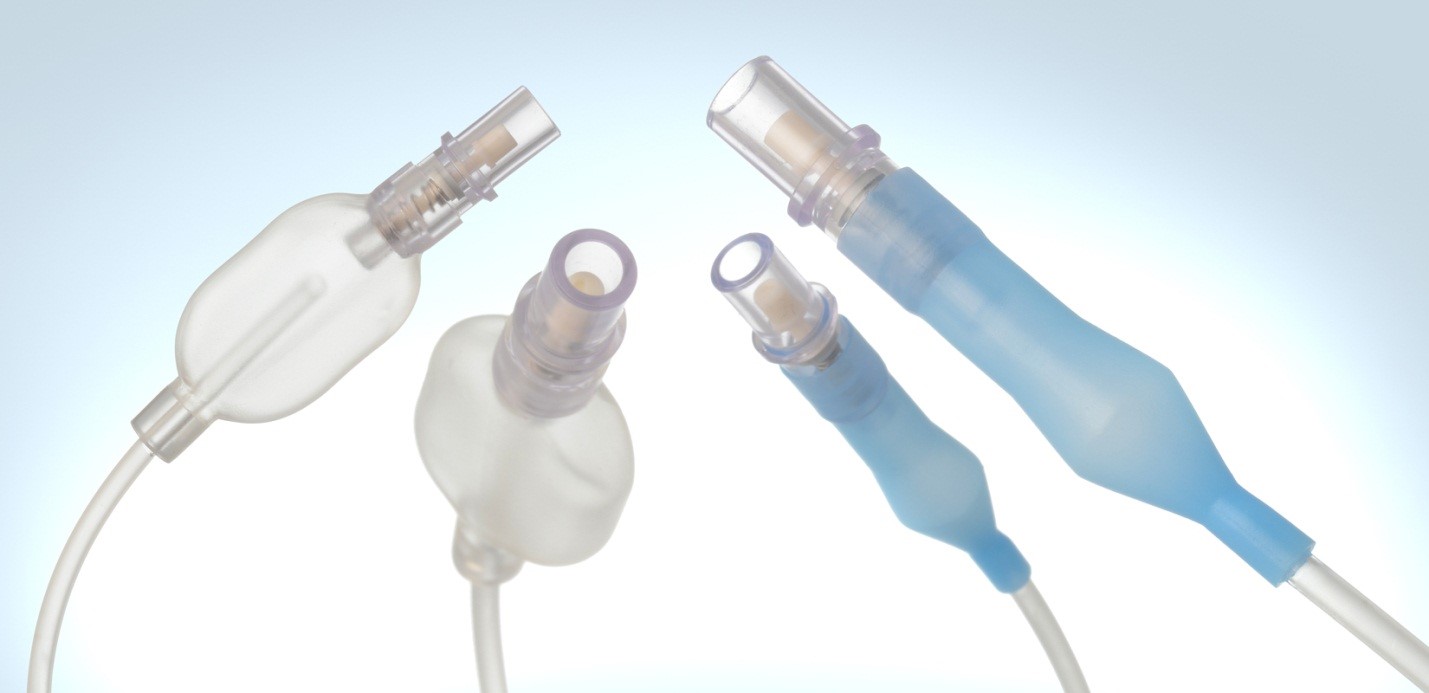
Cannulas. Spring Reinforced Tubing. Y Connectors. Custom Medical Devices.
Blow Molding & Extrusion
Medical Balloons. Airway Management Devices. Multi-Lumen Tubing.
Product Development & Engineering
Medical Device development from ideation stage through prototyping. World Class engineering to optimize both tool and part design.
Get a Quote For Your Project
A Single Source For Your Entire Supply Chain
MDI serves as an integral partner for the world’s largest medical device companies. We’ve brought thousands of devices to market from conception stage all the way through production, assembly, and packaging.
We are a true “single source vendor”. In addition to our several molding facilities, we have a division dedicated exclusively to engineering and design for your new product development and R&D efforts.
We will take your early concept through the design and prototyping stages, early clinical runs, to full volume production and assembly.
After optimizing your device’s production process, we can leverage cost for you into our Mexico facility, where lower cost manufacturing and the ability to ground-ship anywhere in the US gives you a market advantage.
Years Established
Completed Projects
Enhancing the Design of Your Medical Device
Our experienced team of engineers will review your new product concept, or your existing device, and fintie analyses to uncover design optimizations that can be implemented to enhance the efficiency of the manufacturing process. What we’ve been able to do for our partners is quite remarkable, in many cases the reduced scrap and production cycle time savings have produced hundreds of thousands of dollars in annualized savings.

Call For a Quote:
1-800-852-1472
FAQ’s
- What type of clean room molding and assembly do you have?
- Are you registered with the FDA?
- What are the various types of molding services you offer?
- Do you have in-house tooling maintenance?
- Where along our supply chain can you add the most value?
- What kind of quality control systems do you have in place?
- Do you have any plants outside of the US for lower cost labor?
Our Clients
“MDI’s upfront engineering support saved us months of product development.”
“MDI has allowed us to have a single point contact for our Injection molding, Blow molding and assembly needs.”
“Using MDI’s engineering support, took one of our stalled programs from concept to production saving us time and money.”

























HAVE A QUESTION?
WE ARE HERE TO HELP!

